LUMEA Brain Rehab
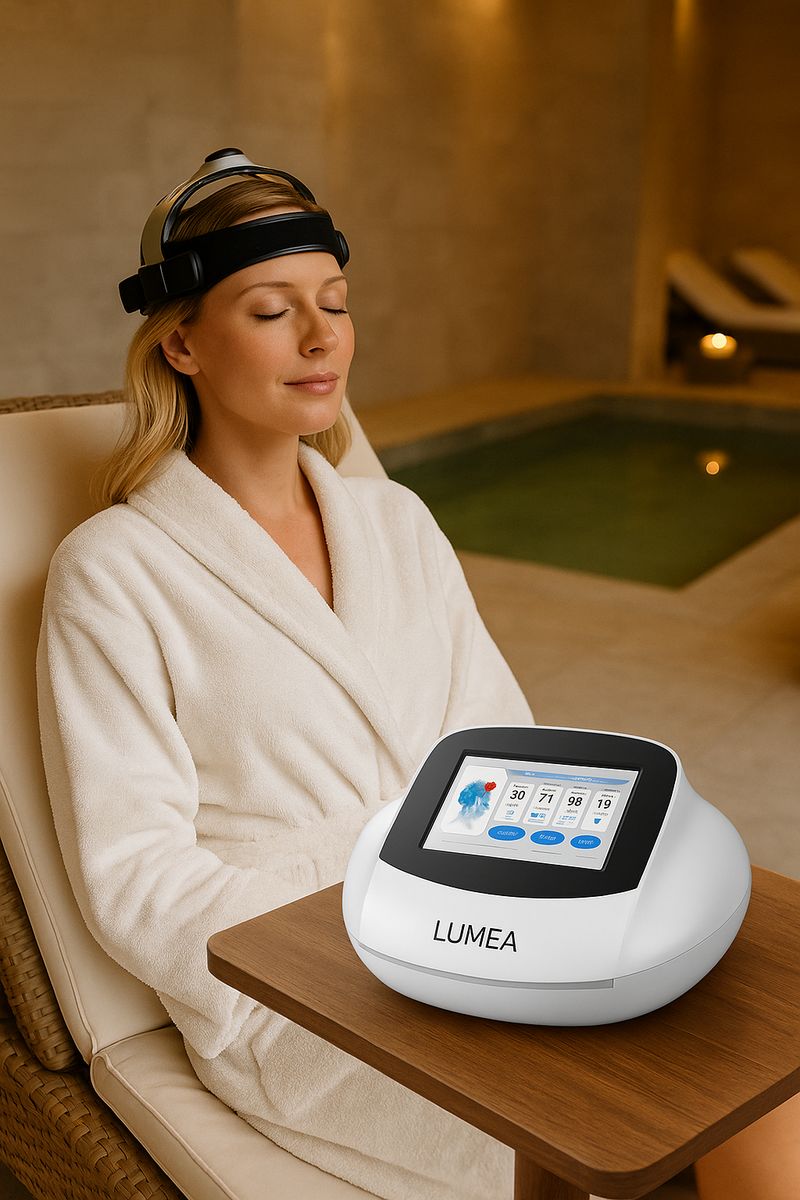
Lumea Brain Rehab – Advanced Non-Invasive Brain Stimulation for Neurological and Mental Health Recovery
Lumea Brain Rehab is a state-of-the-art non-invasive brain stimulation system designed to support brain recovery and functional rehabilitation across a wide range of neurological and psychiatric conditions. Based on the proven principles of repetitive Transcranial Magnetic Stimulation (rTMS), this innovative device uses targeted electromagnetic fields to promote neuroplasticity, improve cerebral blood flow, and restore cognitive and emotional balance—without the need for drugs or invasive procedures.
Mechanism of Action and Core Technology
Lumea Brain Rehab works by delivering alternating electromagnetic fields that safely penetrate the skull and reach deep into the brain tissue. Once inside, these fields generate induced electrical currents that directly stimulate specific brain regions associated with cognitive, emotional, or motor function.
This mechanism activates and modulates neuronal circuits, increases cerebrovascular elasticity, and enhances local blood circulation, creating ideal conditions for neuroregeneration. The system uses a three-dimensional magnetic therapy cap that enables whole-skull synchronous therapy, ensuring even and comprehensive stimulation across multiple brain regions simultaneously—unlike conventional rTMS systems that focus on isolated cortical areas.
User-Centered Design
Ease of use is a key strength of Lumea Brain Rehab. It features an intelligent touchscreen interface, allowing therapists and healthcare professionals to quickly customize sessions and protocols based on the patient’s clinical profile. The interface simplifies the operation without compromising the complexity or effectiveness of the treatment parameters.
The ergonomic therapy cap is designed for optimal comfort and precise positioning, ensuring consistent electromagnetic delivery and minimizing user error. The painless and non-invasive nature of the therapy makes it well-suited for repeated use, including in outpatient, clinical, and integrative health settings.
Applications Across Multiple Clinical Areas
Lumea Brain Rehab has broad application in both neurological rehabilitation and mental health care. It supports recovery and symptom relief in:
Cerebrovascular disorders: Such as stroke, cerebral infarction, Moyamoya disease, migraine, vertigo, aphasia, tinnitus, and post-stroke rehabilitation.
Movement disorders (dyskinesias): Including Parkinson’s disease, dystonia, tremors, and Tourette’s syndrome.
Psychiatric conditions: Including depression, anxiety, sleep disorders, schizophrenia, autism, mania, and neurasthenia.
Brain injury recovery: Useful in cases of concussion, traumatic brain injury (TBI), cerebral hemorrhage recovery, and children’s cerebral palsy.
Its flexibility makes it valuable not only for acute recovery but also for chronic condition management and cognitive enhancement.
Technical Specifications
Lumea Brain Rehab is designed for safe and stable use in a wide variety of healthcare environments:
Ambient temperature range: 5°C – 40°C
Relative humidity: 30% – 75%
Atmospheric pressure range: 700 hPa – 1060 hPa
Power supply: AC 220V ± 22V | Input power: 150VA
Frequency: 50Hz ± 1Hz
Magnetic field intensity options:
Weak mode: 6mT – 10mT
Strong mode: 11mT – 19mT
These technical parameters ensure reliable performance, customizable treatment intensity, and environmental adaptability.
Conclusion
Lumea Brain Rehab brings together advanced rTMS technology, intuitive operation, and a broad therapeutic spectrum to offer a truly comprehensive brain rehabilitation solution. Whether used to treat neurological deficits, support mental health recovery, or optimize cognitive function, it embodies the future of non-invasive neuromodulation. Its precision, safety, and user-friendliness make it an essential tool for modern clinics, rehabilitation centers, and wellness-focused practices committed to brain health and human potential.